Expo
ver canal
ver canal
ver canal
ver canal
ver canal
ver canal
ver canal
ver canal
Química Clínica
HematologíaInmunologíaMicrobiologíaPatologíaTecnologíaIndustria
Eventos
Webinars

- Datos a largo plazo muestran que el cribado con PSA reduce modestamente las muertes por cáncer de próstata
- Prueba de biomarcadores en sangre con marcado CE impulsa el diagnóstico automatizado del Alzheimer
- Ensayo autorizado por la FDA permite pruebas automatizadas integrales de testosterona
- Nanosensor basado en orina monitorea cáncer de pulmón y fibrosis de forma no invasiva
- Prueba de sangre para Alzheimer obtiene marca CE para detectar patología amiloide
- Ensayo de perfilado genómico autorizado por la FDA guía la selección terapéutica en tumores sólidos
- Perfil multiómico ayuda a predecir la respuesta al BCG y la recurrencia en cáncer de vejiga
- Nueva herramienta computacional revela impulsor genético de la neuropatía idiopática
- Firmas específicas del cáncer de mama vinculan la inestabilidad genómica con los resultados clínicos
- Prueba de sangre detecta falta de respuesta temprana en cáncer de próstata metastásico
- Análisis de sangre permite la detección temprana de recaída del mieloma múltiple
- Identifican un “interruptor protector” en el linfoma difuso de células B grandes
- Parámetros rutinarios de análisis de sangre vinculan la anemia con el riesgo de cáncer y la mortalidad
- Nuevo ensayo de función plaquetaria permite monitorizar la terapia antiplaquetaria
- Herramienta pronóstica guía el tratamiento personalizado en cáncer hematológico raro
- Las pruebas en el punto de atención podrían ampliar el acceso al diagnóstico de Mpox
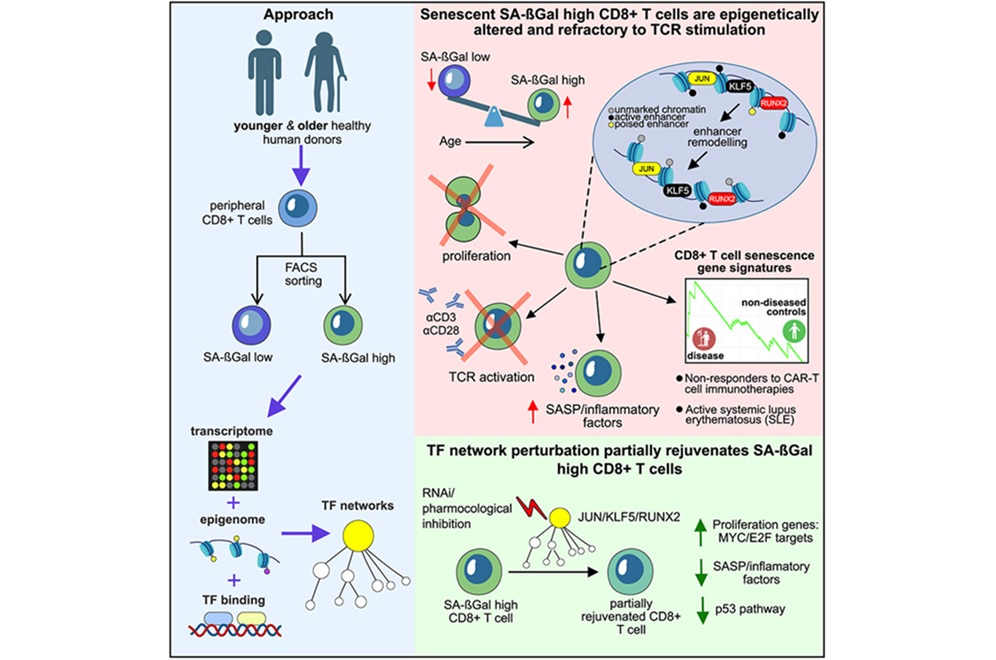
- El perfil de senescencia de las células T podría predecir respuestas a la terapia CAR-T
- Prueba de flujo lateral detecta biomarcadores de sepsis en el punto de atención
- Método de cribado combinado permite identificar casos de lepra en etapas tempranas
- Prueba de anticuerpos en sangre identifica tuberculosis activa y distingue la infección latente
- Vigilancia genómica a gran escala rastrea bacterias resistentes en hospitales europeos
- Biosensor de antígeno detecta tuberculosis activa en una hora
- Panel rápido de susceptibilidad en hemocultivo amplía la cobertura para infecciones por gramnegativos
- Las características del microbioma oral e intestinal permiten identificar el cáncer gástrico temprano
- Un método de microscopía sin marcadores permite una detección más rápida y cuantitativa de la malaria
- Plataforma de 'tumor en un chip' modela la respuesta al tratamiento del cáncer de páncreas
- Nueva plataforma captura vesículas extracelulares para la detección temprana del cáncer
- Un ensayo microfluídico unicelular predice el riesgo de cáncer de mama
- Herramienta de IA predice la no respuesta a terapia dirigida en cáncer colorrectal
- Sistema integrado optimiza el flujo de trabajo preanalítico para pruebas moleculares
- Roche adquirirá PathAI por hasta 1.050 millones de dólares para fortalecer su cartera de diagnósticos con IA
- Seegene presenta plataforma de análisis de datos de PCR en tiempo real
- QuidelOrtho incorpora una plataforma de PCR ultrarrápida con la adquisición de LEX Diagnostics
- Una filial de Roche amplía su cartera de productos ERM con la adquisición de SAGA
- Beckman Coulter obtiene el marcado CE para un ensayo rápido que distingue infecciones bacterianas de virales
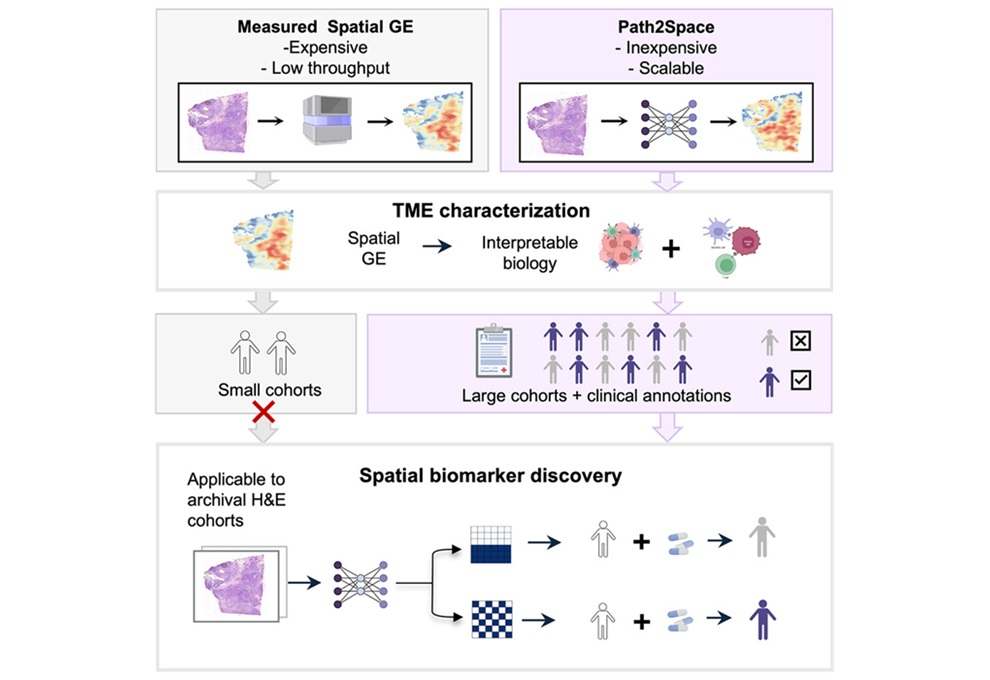
- Herramienta de IA predice la expresión génica espacial del cáncer a partir de imágenes de patología
- Herramienta de patología digital con IA para la estratificación del riesgo en cáncer de mama
- Nueva herramienta de IA revela señales genéticas ocultas en portaobjetos H&E de rutina
- Sistema de IA analiza muestras patológicas rutinarias para predecir resultados del cáncer
- Nuevo enfoque de mapeo tisular identifica una forma de alto riesgo de enfermedad renal diabética

 Expo
Expo
- Datos a largo plazo muestran que el cribado con PSA reduce modestamente las muertes por cáncer de próstata
- Prueba de biomarcadores en sangre con marcado CE impulsa el diagnóstico automatizado del Alzheimer
- Ensayo autorizado por la FDA permite pruebas automatizadas integrales de testosterona
- Nanosensor basado en orina monitorea cáncer de pulmón y fibrosis de forma no invasiva
- Prueba de sangre para Alzheimer obtiene marca CE para detectar patología amiloide
- Ensayo de perfilado genómico autorizado por la FDA guía la selección terapéutica en tumores sólidos
- Perfil multiómico ayuda a predecir la respuesta al BCG y la recurrencia en cáncer de vejiga
- Nueva herramienta computacional revela impulsor genético de la neuropatía idiopática
- Firmas específicas del cáncer de mama vinculan la inestabilidad genómica con los resultados clínicos
- Prueba de sangre detecta falta de respuesta temprana en cáncer de próstata metastásico
- Análisis de sangre permite la detección temprana de recaída del mieloma múltiple
- Identifican un “interruptor protector” en el linfoma difuso de células B grandes
- Parámetros rutinarios de análisis de sangre vinculan la anemia con el riesgo de cáncer y la mortalidad
- Nuevo ensayo de función plaquetaria permite monitorizar la terapia antiplaquetaria
- Herramienta pronóstica guía el tratamiento personalizado en cáncer hematológico raro
- Las pruebas en el punto de atención podrían ampliar el acceso al diagnóstico de Mpox
- El perfil de senescencia de las células T podría predecir respuestas a la terapia CAR-T
- Prueba de flujo lateral detecta biomarcadores de sepsis en el punto de atención
- Método de cribado combinado permite identificar casos de lepra en etapas tempranas
- Prueba de anticuerpos en sangre identifica tuberculosis activa y distingue la infección latente
- Vigilancia genómica a gran escala rastrea bacterias resistentes en hospitales europeos
- Biosensor de antígeno detecta tuberculosis activa en una hora
- Panel rápido de susceptibilidad en hemocultivo amplía la cobertura para infecciones por gramnegativos
- Las características del microbioma oral e intestinal permiten identificar el cáncer gástrico temprano
- Un método de microscopía sin marcadores permite una detección más rápida y cuantitativa de la malaria
- Plataforma de 'tumor en un chip' modela la respuesta al tratamiento del cáncer de páncreas
- Nueva plataforma captura vesículas extracelulares para la detección temprana del cáncer
- Un ensayo microfluídico unicelular predice el riesgo de cáncer de mama
- Herramienta de IA predice la no respuesta a terapia dirigida en cáncer colorrectal
- Sistema integrado optimiza el flujo de trabajo preanalítico para pruebas moleculares
- Roche adquirirá PathAI por hasta 1.050 millones de dólares para fortalecer su cartera de diagnósticos con IA
- Seegene presenta plataforma de análisis de datos de PCR en tiempo real
- QuidelOrtho incorpora una plataforma de PCR ultrarrápida con la adquisición de LEX Diagnostics
- Una filial de Roche amplía su cartera de productos ERM con la adquisición de SAGA
- Beckman Coulter obtiene el marcado CE para un ensayo rápido que distingue infecciones bacterianas de virales
- Encuentran etiologías de COVID prolongada en muestras de sangre con infección aguda
- Dispositivo novedoso detecta anticuerpos contra la COVID-19 en cinco minutos
- Prueba para COVID-19 mediante CRISPR detecta SARS-CoV-2 en 30 minutos usando tijeras genéticas
- Asocian disbiosis del microbioma intestinal con la COVID-19
- Validan prueba rápida novedosa de antígeno para el SARS-CoV-2 con respecto a su exactitud diagnóstica
- Herramienta de IA predice la expresión génica espacial del cáncer a partir de imágenes de patología
- Herramienta de patología digital con IA para la estratificación del riesgo en cáncer de mama
- Nueva herramienta de IA revela señales genéticas ocultas en portaobjetos H&E de rutina
- Sistema de IA analiza muestras patológicas rutinarias para predecir resultados del cáncer
- Nuevo enfoque de mapeo tisular identifica una forma de alto riesgo de enfermedad renal diabética